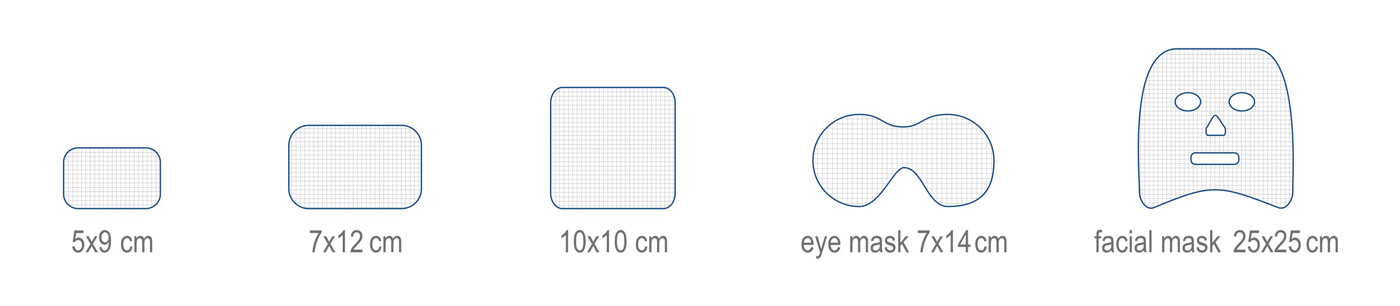
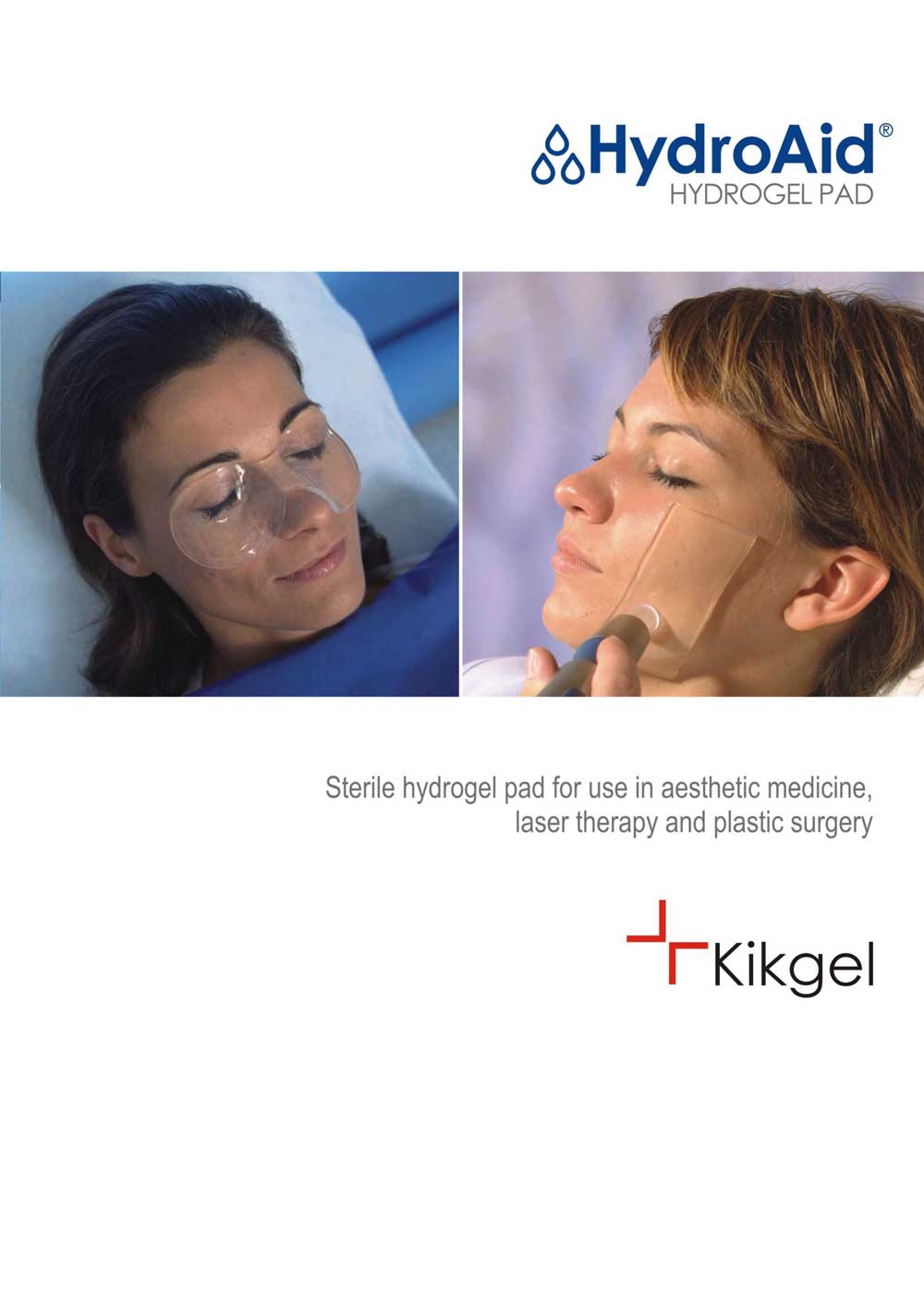
HydroAid®. Hydrogel dressings for aesthetic medicine and plastic surgery
HydroAid® is a modern dressing in the form of a homogenous, transparent, mechanically resistant pad of hydrogel based on a special net of three polymers, including about 90% of water, ca. 3 mm thick. Thanks to this unique structure HydroAid® dressing has many exceptional properties. HydroAid® is applied as a cooling and alleviative hydrogel dressing to protect skin following laser ablation, aesthetic medical procedures, plastic surgery and phlebology. It supports the healing of injured tissue as well.
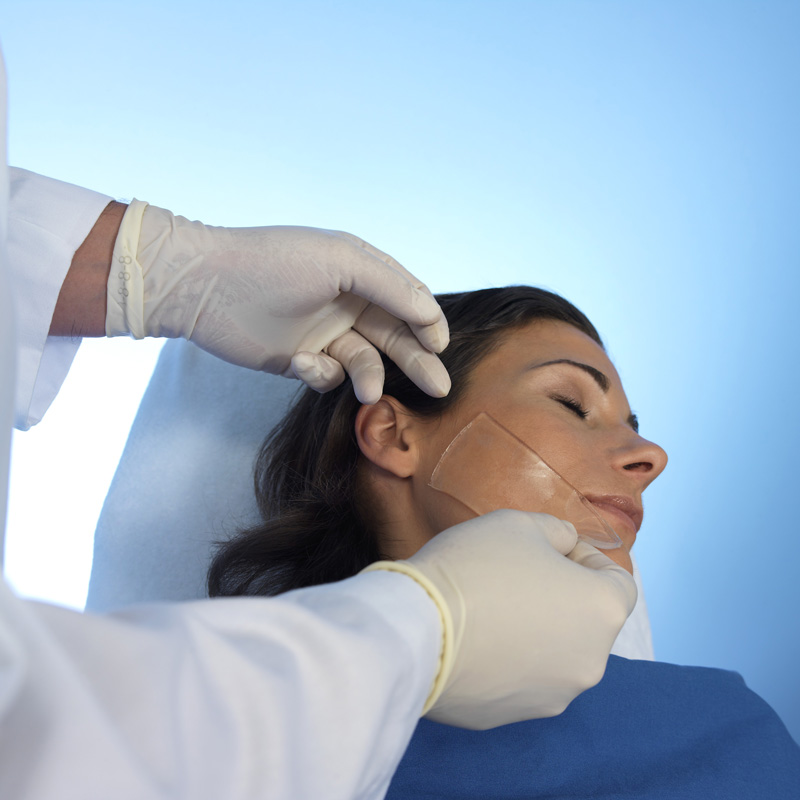
HydroAid® may be used during non-ablative laser procedures, as an elastic and transparent contact layer which cools and protects the epidermis from thermal injuries, thus improving the quality of the procedure and the patient’s level of comfort. It is useful as a coupling agent for ultrasound applications in cases of breached dermis or open wounds, or when a sterile and transparent hydrogel cushion is necessary to improve visibility of examined tissue.

Non-ablative laser procedures
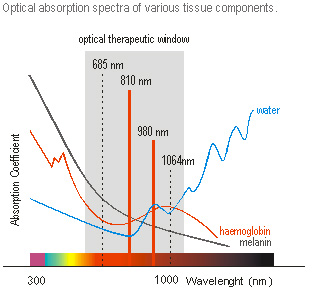
HydroAid® may be used as a cooling hydrogel pad during procedures using lasers of various types (Q-switched, pulsation, half-conductory, KTP) with continuous-wave emission or with intense pulsed light-generating devices (IPL) with wavelengths of 500-1100 nm. In this wavelength range the absorption of laser energy by water contained in the skin and dermis is lower (hydrogel contains mainly water as well), making laser therapy possible for deep levels of tissue through so-called ‘optical therapeutic window’.
Most of all HydroAid® are used during non-ablative laser therapies such as: laser hair removal, tattoos, naevi, hyperpigmentation removal, flat vascular lesion and vascular spider pattern removal.
The use of hydrogel in laser therapies moisturises the corneous layer of the epidermis very well, by preventing dispersal of the laser beam and its reflection on the surface of the skin.
The use of hydrogel during surgical laser procedures eliminates excess steam and smoke from coagulated tissues.
During laser hair removal through hydrogel dressing, the laser head touches only the sterile hydrogel layers, thanks to which it is kept clean; no tissues or hair remnants collect on its surface, which subsequently exerts a positive influence on the procedure’s hygiene, quality and safety.
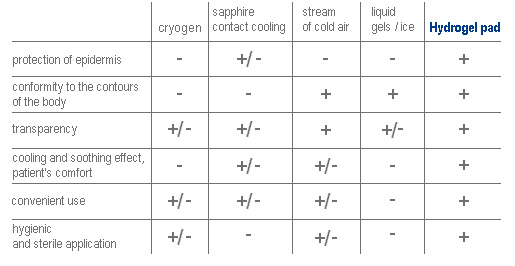
Hydrogel pads have many advantages in comparison with other cooling methods used in non-ablative procedures, according to Julian M. Handley “Adverse events associated with nonablative cutaneous visible and infrared laser treatment”. Journal of the American Academy of Dermatology, September 2006;55,3:482-489.
Laser ablation procedures
Laser ablation procedures, due to the great volume of energy transmitted to tissues, may cause undesirable complications such as erythema, oedema, hyperpigmentation, hypertrophy scars, and increased skin sensitivity.
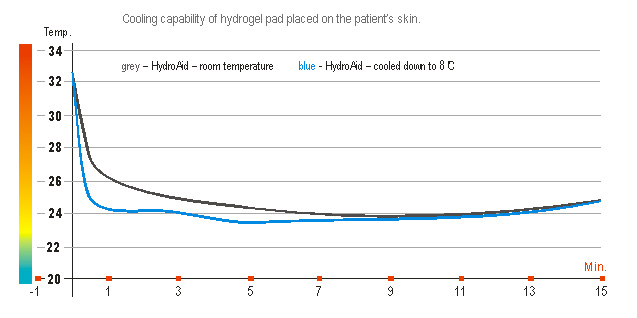
Using HydroAid® dressing, thanks to its great heat-absorbing capacity, cools the area subjected to therapy. Thanks to that the painfulness of the procedure itself and the risk of burns or other skin injuries occurrence are minimised. The dressing produces a pleasant, soothing feeling, making the procedure itself comfortable for the patient.
The hydrogel pad not only ensures appropriate thermal balance but also moisture balance on the injuried skin surface, which, in turn, accelerates epithelialization and tissue healing processes.
Hydrogel pads have many advantages in comparison with other cooling methods used in non-ablative procedures, according to Julian M. Handley, ‘Adverse events associated with nonablative cutaneous visible and infrared laser treatment’, Journal of the American Academy of Dermatology, September 2006; 55, 3: 482-489
Use in the aesthetic medicine
HydroAid® pads may be used after microdermabrasion therapy, peeling, face lifting, and depigmentation therapy. Most of these procedures consist of chemical and mechanical epidermal desquamation which causes reddening of the areas subjected to the therapy, as well as occasional oedema and burning sensations.
The use of HydroAid® after such procedures cools and moisturises the irritated tissues. Epidermal regeneration processes are accelerated and the patient feels relief and the soothing effect of the hydrogel dressing.
Mesotherapy and face lifting are methods of treatment for various skin diseases and also of improvement of appearance based on injecting small amounts of healing substances directly into lesions. Following such procedures, erythema and other undesirable reactions often occur. To alleviate them, the use of HydroAid® hydrogels is recommended. Needle injections of e.g. botox, hyaluronic acid and other fillers can also be conducted through HydroAid® dressing, which is transparent and enables observation of the area undergoing therapy.
HydroAid® as a cooling and soothing hydrogel pad is also used in photo-rejuvenating procedures (small blood vessels, small hyperpigmentation reduction) with the use of IPL or VPL pulsation lamps, or after thermolifting or thermage procedures with the use of ultrasonic waves.
Use in the phlebology
HydroAid® may be applied in certain phlebology procedures, such as closure of veins using laser energy (EVLT) or steam (SVS).
Because such procedures result in increased temperatures in the tissues themseves, it is necessary to use alleviating agents during or after the procedures. HydroAid® is perfect for this.
To intensify the cooling and soothing effect before the start of the procedure, HydroAid® dressings can be cooled in a refrigerator.
Use in ultrasonography
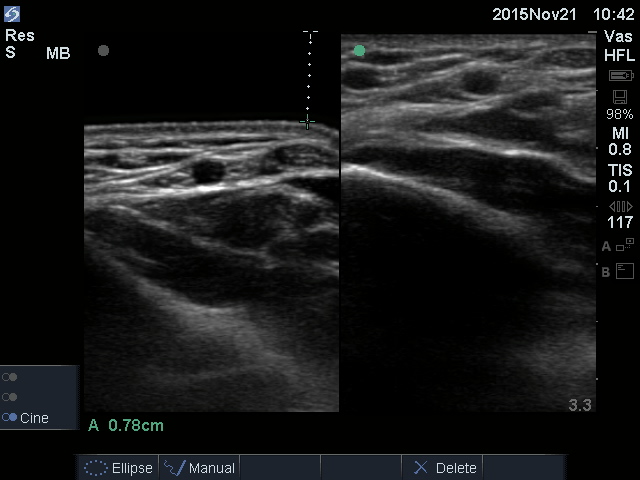
HydroAid® may also be used as a substitute for ultrasound gel during special ultrasound examinations where the use of liquid gels is not possible. With sterile HydroAid® products, it is possible to conduct examinations on open wounds, e.g. Doppler examinations of ulcers or examinations of third-degree burns, in order to determine the depth of necrosis. HydroAid® is also used in regions where it is difficult to conduct local examinations (e.g. tendons, small joint injuries, carpal tunnel syndrome, fontanelle) or where the sterile and transparent coupling agent between the wound and the instrument head is necessary to improve the visibility of surface structures.
HydroAid® pad, thanks to its dense and mechanically durable hydrogel structure, is an excellent conductor of ultrasound energy.
HydroAid®, especially in width of 6 mm and 10 mm, is used in ultrasound screening in which keeping a distance is necessary to improve the visibility of structures located near the surface or in areas of irregular shape and posing difficulties in spot examination.
It is especially useful in examination of :
- surface blood vessels
- tendons
- small joint injuries
- fontanels
- bony prominences
- carpal tunnel syndrome
- lymph nodes
- thyroid gland
- superficial breast cancer
- the optic nerve as the assessment of intracranial pressure
HydroAid® is applied as a sterile protector of skin and wounds and can be used as a scanning barrier between a transducer and a patient’s skin in the course of ultrasound diagnostics. With a sterile HydroAid® hydrogel pad, it is possible to examine the open wounds where the use of liquid gels is not possible, e.g.:
- renal transplant wounds,
- Doppler examinations of ulcers,
- examination of third-degree burns to determine depth of necrosis
Use in plastic surgery
HydroAid dressings are used in plastic surgery mainly after invasive procedures on the skin as cooling, soothing and sterile dressings.
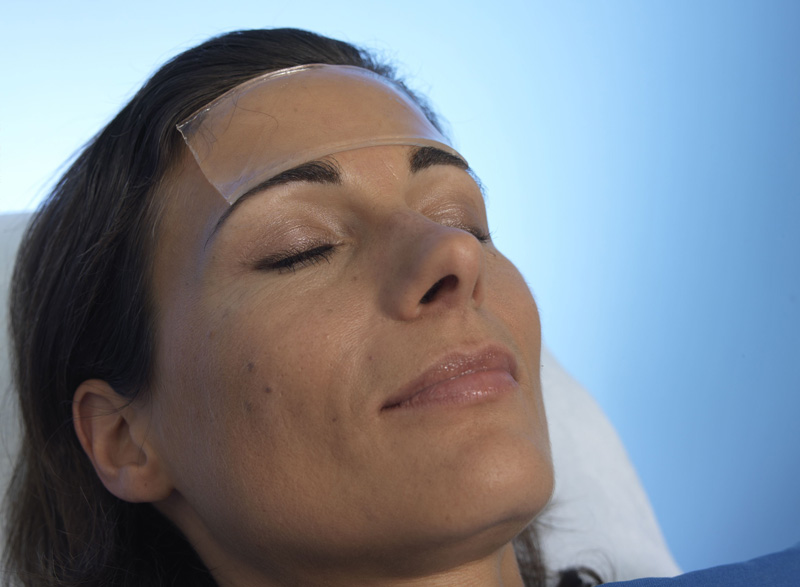
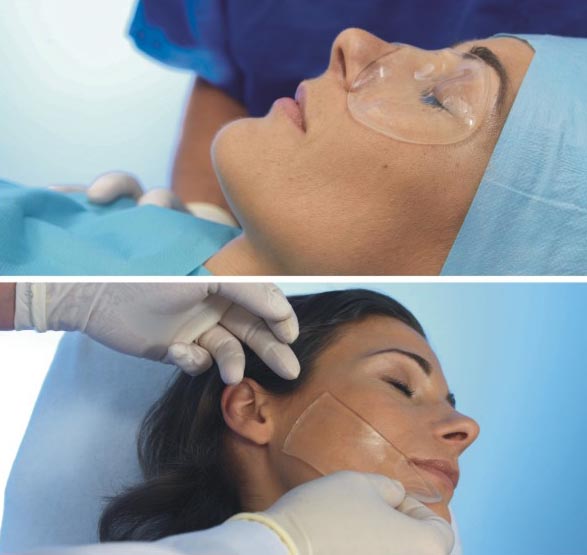
The dressing may be used to protect wounds after surgical suturing and after laser liposuction procedures, while a mask-shaped dressing on the eyes may be used following blepharoplasty procedures (surgical procedures on the eyelids).
HydroAid dressing features absorption properties so that blood or exudate are absorbed into hydrogel. The dressing’s exudate absorption capacity is 20 ml per 100 square centimetres of wound surface. Its transparent structure enables observation of the area subjected to a procedure without removing the dressing.
How to use
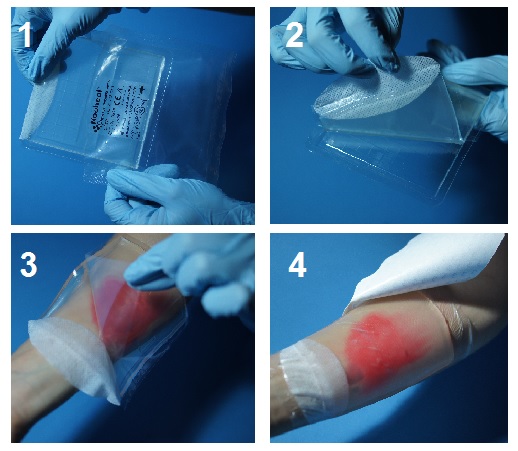
The HydroAid® dressing is placed in a mould, covered with a leaf of foil and hermetically packed in a foil bag. The piece of fibre and foil, which adheres to the surface, facilitates the removal of the dressing what is presented on figures 1-4.
To intensify the cooling and soothing effect before the start of the procedure, HydroAid® dressings can be cooled in a refrigerator, but it should not be frozen completely. Should the patient experience some “overheating” of the treated skin, do not hesitate to remove the hydrogel pad and to cool it down on the “cold pack” cushion.
Removing and application of the Eye Mask hydrogel pad.
To Download
Indications of hydrogel pads in laser therapy procedures are presented in following publications:
- Dew Point Effect of Cooled Hydrogel Pads on Human Stratum Corneum Biosurface, Emmanuelle Xhauflaire-Uhoda, Philippe Paquet, Gérald E. Piérard, Dermatology 2008; 216:37-39.
- Right-left comparison study of hydrogel pad versus transparent fluid gel in patients with dermo-cosmetic lesions undergoing non-ablative laser therapy. Cassuto D, Mollia JF, Scrimali L, Sirago P. Cosmet Laser Ther. 2009 Mar;11(1):45-51.
- Reduction in pain and erythema by an innovative hydrogel pad. Sibylle Meier, Reinhard Dummer, Hautnah Dermatologie 4/05.
- Julian M. Handley, „Adverse events associated with nonablative cutaneous visible and infrared laser treatment”, Journal of the American Academy of Dermatology, September 2006; 5, 3: 482-489.
Symbols

Batch code
Indicates the manufacturer’s batch code so that the batch or lot can be identified.

Use-by date
Indicates the date after which the medical device is not to be used.

Manufacturer
Indicates the medical device manufacturer, asdefined in EU Directives: 90/385/EEC, 93/42/EEC and 98/79/EC.
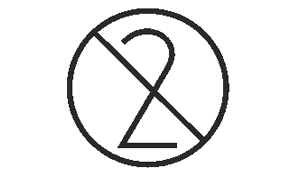
Do not re-use
Indicates a medical device that is intended for one use, or for use on a single patient during a single procedure.
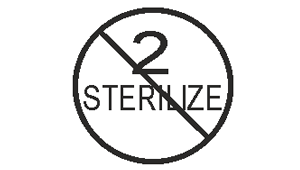
Do not resterilize
Indicates a madical device that is not to be resterilized.

Sterilized using irradiation
Indicates a medical device that has been sterilized using irradiation.

Keep dry
Indicates a medical device that needs to be protected from moisture.

Temperature limit
Indicates the temperature limits to which the medical device can be safely exposed.
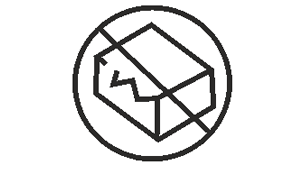
Do not use if package is damaged
Indicates a medical device that should not be used if the package has been damaged or opened.

Keep clean
It means that after use, the packaging should be discarded in a suitable container so that it does not endanger or litter the environment.

CE mark
Indicates conformity with health, safety, and environmental protection standards for products sold within the European Economic Area (EEA)

Consult instructions for use
Indicates the need for the user to consult the instructions for use.